The spinal cord possesses precise neural circuitry to transmit messages between the brain and the body. Among the core activities of the body, there are two essential functions provided by the spinal cord: process somatosensory information and execute movement. Besides those essential functions, the spinal cord also manages reflex actions, physical coordination, and rhythmic motor activity.
Different neural progenitor subtypes are specified and differentiate into diverse neuronal/ glial cells to form an ordered neural network during spinal cord development. Gaining further insight into the temporal and cellular features of developing spinal cord is critical for advancing the regeneration therapy strategies for spinal cord diseases.
In a study published in EMBO Reports (DOI:10.15252/embr.202152728), a research team led by Prof. DAI Jianwu from the Institute of Genetics and Developmental biology of the Chinese Academy of Sciences, in collaboration with Prof. HU Yali from Nanjing university medical school, revealed the developmental timeline and transcriptional heterogeneity in developing neural cells during embryonic and fetal stages of human spinal cord development.
The researchers identified transcriptional characteristics and revealed the composition of neural populations at various stages by performing both nuclear single-cell RNA sequencing and cellular single-cell RNA sequencing of the developing spinal cord. In human spinal cord, the increase of neuronal cells ceases before gestational week (GW) 14. The majority of ventral neurons appear earlier than GW7, while the dorsal neurons mostly produced after GW7. Astrocyte development starts at GW8 and gradually stops before GW23; in contrast, oligodendrocyte development was detected from GW8 onwards and continued to increase with advancing fetal age.
Besides, researchers aligned human data (GW7–11) with mouse developing spinal cord transcriptomics data (E9.5–13.5) to investigate the species difference of spinal cord development. The results showed several human neuronal subclasses lacked mouse homologues, and human spinal cord undergoes a more extended gliogenesis period during fetal development than rodents.
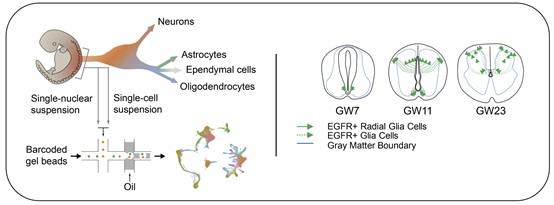
Finally, researchers revealed a group of EGFR-expressing transitional glial cells. These cells may arise from neuronal progenitor cells during the later neurogenesis stage and displayed a markedly spatiotemporal arrangement feature during human spinal cord development. Intercellular communication network assay indicates that these EGFR-expressing glial cells functional interact with other glial cells through Notch and EGFR signaling pathway during development.
This study outlined the landscape of neural development in the human spinal cord. Transcriptome characteristics of neural cell types present at various stages of development were defined at single-cell resolution. It provides fundamental insights into human spinal cord development and may inform cellular therapy approaches to treat spinal cord diseases.
Schematic diagrams showed the single-cell sequencing process (left) and EGFR-expressing cells during spinal cord development (right). (Image by IGDB)
Contact:
Dr. DAI Jianwu
Institute of Genetics and Developmental biology, Chinese Academy of Sciences
 Schematic diagrams showed the single-cell sequencing process (left) and EGFR-expressing cells during spinal cord development (right). (Image by IGDB)Contact:Dr. DAI JianwuInstitute of Genetics and Developmental biology, Chinese Academy of SciencesEmail: jwdai@genetics.ac.cn
Schematic diagrams showed the single-cell sequencing process (left) and EGFR-expressing cells during spinal cord development (right). (Image by IGDB)Contact:Dr. DAI JianwuInstitute of Genetics and Developmental biology, Chinese Academy of SciencesEmail: jwdai@genetics.ac.cn CAS
CAS
 中文
中文




.png)
