Prime editing (PE) is a newly described precision genome editing technology capable of generating precise base substitutions, deletions, or insertions of DNA bases at a specified target site in the genome of living cells.
In 2020, GAO Caixia's team from the Institute of Genetics and Developmental Biology (IGDB), Chinese Academy of Sciences, successfully led the first establishment and optimization of using prime editing in rice and wheat through exploring the fusion of different reverse transcriptase enzymes, best experimental temperatures, pegRNA expression methods, PBS (Primer binding site sequence) and RT (Reverse transcriptase) template sequence lengths and other conditions. Nevertheless, the editing efficiency of prime editors for use in plants remained low and seemed to be highly target dependent. These shortcomings severely limited the utility of prime editing in plants.
Recently, GAO's group and collaborators developed a more efficient and improved plant prime editing system, named ePPE (Engineered Plant Prime Editor). Different from previously reported methods to improve the editing efficiency of prime editing through mainly optimizing the pegRNA, this study focused on the modification of the protein components in the prime editor.
The researchers found that the deletion of the RNase H domain in the M-MLV RT or fusion of a viral nucleocapsid (NC) protein at the N-terminus of the M-MLV RT greatly enhanced overall prime editing efficiencies in plant cells by 2.0-fold or 3.2-fold, respectively.
Furthermore, the researchers combined both approaches to develop the enhanced engineered plant prime editor, or ePPE. Compared with the original PPE, ePPE can increase editing efficiencies by an average of 5.8-fold when evaluating a variety of editing types, such as base substitutions, small fragment insertions, deletions, and insertions or deletions of larger fragments, without increasing the occurrence of off-target editing events or editing by-products.
When combining the newly developed ePPE with the dual-pegRNA strategy previously reported by GAO’s group and the epegRNA (Engineered pegRNA) strategy developed by David Liu's group in 2021, the efficiency of the prime editing system for generating precise editing events at endogenous genes can be further improved.
Finally, the researchers used the ePPE system to successfully generate new rice plants that are resistant to two different herbicides, imazamox nicotinic acid and nicosulfuron.
In summary, they successfully developed a new prime editing system, ePPE, that can drastically improve prime editing efficiencies in plants through engineering the protein components of PPE, and demonstrated that the combination of ePPE with pegRNA optimization strategies can further improve the editing efficiency across a variety of cell types.
This new editing system is expected to expand the utility of prime editing in agricultural breeding, crop improvement and more.
CAO Xiaofeng's group from the Institute of Genetics and Developmental Biology of the Chinese Academy of Sciences and HUANG Xingxu's research group from Shanghai Tech University jointly participated in the research.
This research was supported by the National Natural Science Foundation of China and the Strategic Pilot Project A of the Chinese Academy of Sciences.
Figure. The establishment and application of ePPE, an efficient new genome editor for use in plants. a-b, Evaluating the original PPE with new prime editing variants using a fluorescent assay. c, Overall editing frequencies induced by PPE, PPE-ΔRNase H, PPE-NC-v1 and PPE-NC-v2. d, Schematic diagram of ePPE. e, Overall editing frequencies induced by PPE, PPE-ΔRNase H, PPE-NC-v1 and ePPE. f, Comparison of editing efficiencies of different PPEs with epegRNAs or dual-pegRNA strategy. g, Phenotype of an ALS-W548M rice mutant under different herbicide treatments. (Image by IGDB)
Contact:
Dr. GAO Caixia
Institute of Genetics and Developmental Biology, Chinese Academy of Sciences
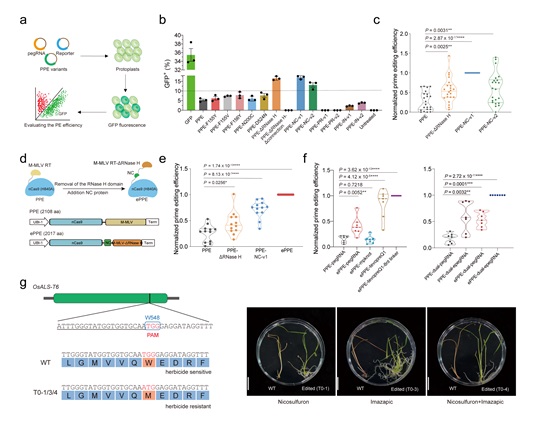 Figure. The establishment and application of ePPE, an efficient new genome editor for use in plants. a-b, Evaluating the original PPE with new prime editing variants using a fluorescent assay. c, Overall editing frequencies induced by PPE, PPE-ΔRNase H, PPE-NC-v1 and PPE-NC-v2. d, Schematic diagram of ePPE. e, Overall editing frequencies induced by PPE, PPE-ΔRNase H, PPE-NC-v1 and ePPE. f, Comparison of editing efficiencies of different PPEs with epegRNAs or dual-pegRNA strategy. g, Phenotype of an ALS-W548M rice mutant under different herbicide treatments. (Image by IGDB)Contact:Dr. GAO CaixiaInstitute of Genetics and Developmental Biology, Chinese Academy of SciencesEmail: cxgao@genetics.ac.cn
Figure. The establishment and application of ePPE, an efficient new genome editor for use in plants. a-b, Evaluating the original PPE with new prime editing variants using a fluorescent assay. c, Overall editing frequencies induced by PPE, PPE-ΔRNase H, PPE-NC-v1 and PPE-NC-v2. d, Schematic diagram of ePPE. e, Overall editing frequencies induced by PPE, PPE-ΔRNase H, PPE-NC-v1 and ePPE. f, Comparison of editing efficiencies of different PPEs with epegRNAs or dual-pegRNA strategy. g, Phenotype of an ALS-W548M rice mutant under different herbicide treatments. (Image by IGDB)Contact:Dr. GAO CaixiaInstitute of Genetics and Developmental Biology, Chinese Academy of SciencesEmail: cxgao@genetics.ac.cn CAS
CAS
 中文
中文




.png)
