Apart from the established roles in bioenergetics and biosynthesis, mitochondria are signaling organelles that communicate their fitness to the nucleus, triggering transcriptional programs to adapt homeostasis stress that is essential for organismal health and aging. While there are several major regulators involved in mito-nuclear communication under stress conditions, the underlying mechanisms are remained to be explored.
In a review article of Trends in Biochemical Sciences, Dr. TIAN Ye's group from the Institute of Genetics and Developmental Biology, the Chinese Academy of Sciences, summarized recent studies on the mechanisms by which mito-nuclear communication modulates epigenomes and their effects on regulating the aging process. Insights into understanding how mitochondrial metabolites serve as pro-longevity signals and how aging affects this communication will help us develop interventions to promote longevity and health. This study entitled “Mitochondrial-to-nuclear Communication in Aging: An Epigenetic Perspective” has been published on April 6th (https://doi.org/10.1016/j.tibs.2022.03.008).
Firstly, they summarized that mitochondrial metabolites can serve as substrate or cofactors to mediate epigenetic modifications, such as histone methylation and histone acetylation. Furthermore, this review deeply explores the effects of multiple mitochondrial metabolites on aging regulation. They discuss examples of mitochondrial metabolites, including acetyl-CoA, alpha-ketoglutarate (α-KG), NAD+, and methionine, as potential longevity regulators and how changes in their abundance influence the epigenomes and the aging process in different organisms. Given that the metabolic status of the cell not only changes during aging but is also susceptible to environmental stimuli, they discussed how mitochondrial stress signals modulate aging via epigenetic regulations. Future studies will help us develop interventions on mitochondrial-to-nuclear communications to mimic the beneficial epigenetic changes for delaying aging or alleviating age-onset diseases.
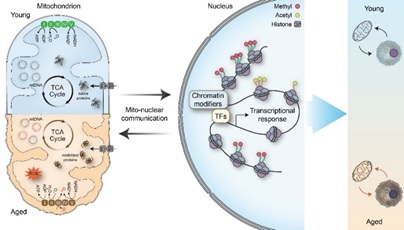
Figure. Mito-nuclear communication and aging (Image by IGDB)
This work is supported by the National Key R&D Program of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, and the National Natural Science Foundation of China.
Contact:
Dr. TIAN Ye
Institute of Genetics and Developmental Biology, the Chinese Academy of Sciences
 Figure. Mito-nuclear communication and aging (Image by IGDB)This work is supported by the National Key R&D Program of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, and the National Natural Science Foundation of China.Contact:Dr. TIAN YeInstitute of Genetics and Developmental Biology, the Chinese Academy of SciencesEmail: ytian@genetics.ac.cn
Figure. Mito-nuclear communication and aging (Image by IGDB)This work is supported by the National Key R&D Program of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, and the National Natural Science Foundation of China.Contact:Dr. TIAN YeInstitute of Genetics and Developmental Biology, the Chinese Academy of SciencesEmail: ytian@genetics.ac.cn CAS
CAS
 中文
中文




.png)
